[ad_1]
Summary
Background
G protein–coupled receptor, household C, group 5, member D (GPRC5D) is an orphan receptor expressed in malignant plasma cells. Talquetamab, a bispecific antibody in opposition to CD3 and GPRC5D, redirects T cells to mediate killing of GPRC5D-expressing myeloma cells.
Strategies
In a section 1 examine, we evaluated talquetamab administered intravenously weekly or each different week (in doses from 0.5 to 180 μg per kilogram of physique weight) or subcutaneously weekly, each different week, or month-to-month (5 to 1600 μg per kilogram) in sufferers who had closely pretreated relapsed or refractory a number of myeloma that had progressed with established therapies (a median of six earlier traces of remedy) or who couldn’t obtain these therapies with out unacceptable uncomfortable side effects. The first finish factors — the frequency and sort of dose-limiting poisonous results (examine half 1 solely), antagonistic occasions, and laboratory abnormalities — had been assessed with a view to choose the beneficial doses for a section 2 examine.
Outcomes
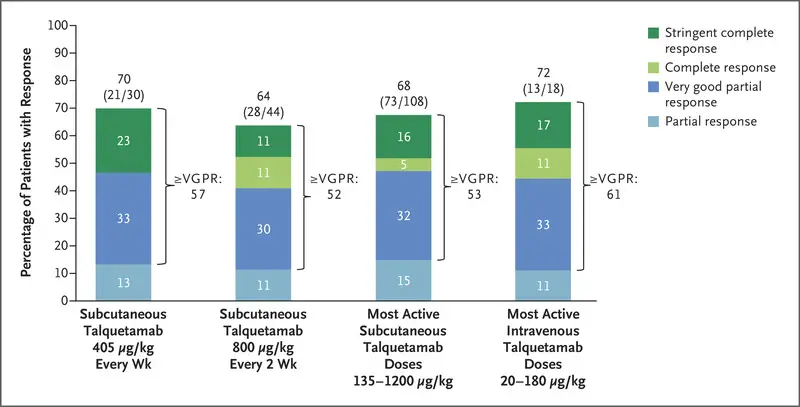
On the data-cutoff date, 232 sufferers had acquired talquetamab (102 intravenously and 130 subcutaneously). On the two subcutaneous doses beneficial for a section 2 examine (405 μg per kilogram weekly [30 patients] and 800 μg per kilogram each different week [44 patients]), frequent antagonistic occasions had been cytokine launch syndrome (in 77% and 80% of the sufferers, respectively), skin-related occasions (in 67% and 70%), and dysgeusia (in 63% and 57%); all however one cytokine launch syndrome occasion had been of grade 1 or 2. One dose-limiting poisonous impact of grade 3 rash was reported in a affected person who had acquired talquetamab on the 800-μg dose degree. At median follow-ups of 11.7 months (in sufferers who had acquired talquetamab on the 405-μg dose degree) and 4.2 months (in those that had acquired it on the 800-μg dose degree), the chances of sufferers with a response had been 70% (95% confidence interval [CI], 51 to 85) and 64% (95% CI, 48 to 78), respectively. The median period of response was 10.2 months and seven.8 months, respectively.
Conclusions
Cytokine launch syndrome, skin-related occasions, and dysgeusia had been frequent with talquetamab remedy however had been primarily low-grade. Talquetamab induced a considerable response amongst sufferers with closely pretreated relapsed or refractory a number of myeloma. (Funded by Janssen Analysis and Growth; MonumenTAL-1 ClinicalTrials.gov quantity, NCT03399799.)
[ad_2]
Source link