[ad_1]
Summary
Background
In a multinational, part 3, head-to-head trial, ibrutinib, a Bruton’s tyrosine kinase (BTK) inhibitor, was in contrast with zanubrutinib, a BTK inhibitor with higher specificity, as therapy for relapsed or refractory persistent lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). In prespecified interim analyses, zanubrutinib was superior to ibrutinib with respect to general response (the first finish level). Knowledge from the ultimate evaluation of progression-free survival at the moment are out there.
Strategies
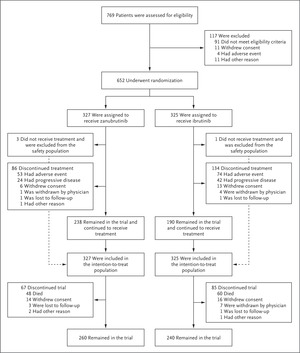
We randomly assigned, in a 1:1 ratio, sufferers with relapsed or refractory CLL or SLL who had obtained at the very least one earlier course of remedy to obtain zanubrutinib or ibrutinib till the prevalence of illness development or unacceptable poisonous results. On this last evaluation, progression-free survival (a key secondary finish level) was assessed with the usage of a hierarchical testing technique to find out whether or not zanubrutinib was noninferior to ibrutinib. If noninferiority was established, the prevalence of zanubrutinib was assessed and claimed if the two-sided P worth was lower than 0.05.
Outcomes
At a median follow-up of 29.6 months, zanubrutinib was discovered to be superior to ibrutinib with respect to progression-free survival amongst 652 sufferers (hazard ratio for illness development or loss of life, 0.65; 95% confidence interval, [CI], 0.49 to 0.86; P=0.002), as assessed by the investigators; the outcomes had been just like these as assessed by an independent-review committee. At 24 months, the investigator-assessed charges of progression-free survival had been 78.4% within the zanubrutinib group and 65.9% within the ibrutinib group. Amongst sufferers with a 17p deletion, a TP53 mutation, or each, those that obtained zanubrutinib had longer progression-free survival than those that obtained ibrutinib (hazard ratio for illness development or loss of life, 0.53; 95% CI, 0.31 to 0.88); progression-free survival throughout different main subgroups persistently favored zanubrutinib. The proportion of sufferers with an general response was larger within the zanubrutinib group than within the ibrutinib group. The security profile of zanubrutinib was higher than that of ibrutinib, with fewer opposed occasions resulting in therapy discontinuation and fewer cardiac occasions, together with fewer cardiac occasions resulting in therapy discontinuation or loss of life.
Conclusions
In sufferers with relapsed or refractory CLL or SLL, progression-free survival was considerably longer amongst sufferers who obtained zanubrutinib than amongst those that obtained ibrutinib, and zanubrutinib was related to fewer cardiac opposed occasions. (Funded by BeiGene; ALPINE ClinicalTrials.gov quantity, NCT03734016.)
[ad_2]
Source link