[ad_1]
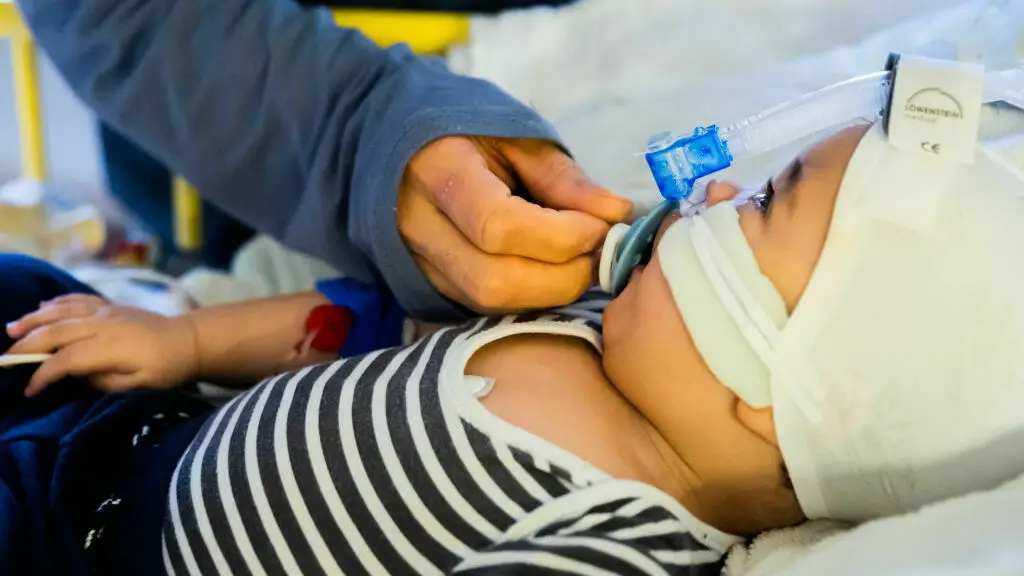
Every year, RSV infections ship as much as 80,000 youngsters beneath 5 to the hospital for emergency therapy. A brand new antibody therapy may shield the youngest youngsters — newborns and up infants as much as 2 years outdated.
Christoph Soeder/dpa/image alliance by way of Getty I
disguise caption
toggle caption
Christoph Soeder/dpa/image alliance by way of Getty I
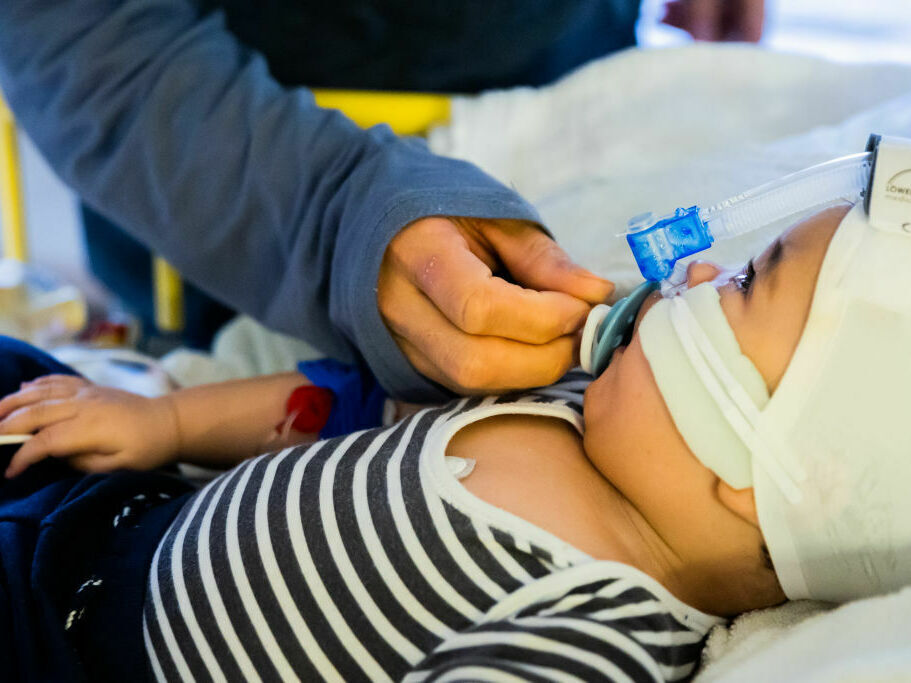
Every year, RSV infections ship as much as 80,000 youngsters beneath 5 to the hospital for emergency therapy. A brand new antibody therapy may shield the youngest youngsters — newborns and up infants as much as 2 years outdated.
Christoph Soeder/dpa/image alliance by way of Getty I
Cheryl Meany, a highschool trainer from Camillus, N.Y., was excited when she discovered she was carrying twins in 2014. However her pleasure shortly turned to fret as medical doctors flagged a number of well being issues, together with attainable mind lesions.
So she wanted a second to course of when her husband, a respiratory therapist, proposed enrolling the soon-to-be-born infants in an experimental examine for an unrelated sickness. It was a trial for a protecting therapy for RSV or respiratory syncytial virus, a typical respiratory virus that may be fairly extreme in younger kids.
“It took me aback, like ‘What are you even speaking about? I do not even know what you are asking me proper now,'” Meany mentioned.
That was in 2014, a number of years earlier than the recent RSV surge overwhelmed hospitals throughout the nation. However Meany was fearful in regards to the sickness again then after seeing a few of her buddies’ youngsters find yourself within the hospital from it. Up to 80,000 children beneath 5 are admitted for RSV annually.
So she enrolled her daughters within the trial for a monoclonal antibody that works to stop RSV-induced decrease respiratory tract an infection in infants. Her resolution helped transfer ahead some of the promising therapies to guard infants from extreme impacts of RSV in many years.
In January, drugmakers AstraZeneca and Sanofi announced the U.S. Meals and Drug Administration is formally reviewing their software to get the therapy – referred to as nirsevimab – authorized within the U.S., together with outcomes from the trial the Meany twins joined.
AstraZeneca mentioned its third part trial outcomes confirmed its single-dose therapy was almost 75% effective at stopping extreme an infection in infants all through an RSV season. The info was printed in March 2022 within the New England Journal of Medicine.

Earlier than they had been born in 2015, twins, Stella and Cassidy Meany (left to proper), had been enrolled in a trial for a preventative therapy for RSV. The therapy might quickly be out there to guard newborns towards the respiratory sickness.
Cheryl Meany
disguise caption
toggle caption
Cheryl Meany

Earlier than they had been born in 2015, twins, Stella and Cassidy Meany (left to proper), had been enrolled in a trial for a preventative therapy for RSV. The therapy might quickly be out there to guard newborns towards the respiratory sickness.
Cheryl Meany
Dr. William Schaffner, medical director on the Nationwide Basis for Infectious Illnesses who was not concerned on this analysis, mentioned the outcomes recommend nirsevimab may considerably scale back the numbers of infants which might be hospitalized annually for RSV.
“The potential impression in assuring a wholesome infancy for a really massive proportion of the infants born right here in the USA — and even past — is doubtlessly very, very massive,” Schaffner mentioned.
A type of ‘passive immunization’
The drug – a long-lasting antibody injection – is meant for newborns or different infants dealing with their first RSV season, and for infants as much as 24 months of age of their second RSV season, in accordance with AstraZeneca’s press launch.
Dr. Joseph Domachowske, a pediatric infectious illness specialist at Upstate Medical College Hospital in Syracuse, helped launch the earliest part of the nirsevimab examine.
“RSV is the primary motive why infants and younger kids are hospitalized, not simply within the U.S., however internationally,” he mentioned.
He defined that the antibody therapy shouldn’t be a vaccine however is what scientists check with as “passive immunization.” The antibodies towards RSV flow into within the infants’ our bodies defending towards the virus, ought to the kid be uncovered.
“It would not trigger an immune response itself,” he mentioned, and it additionally would not trigger the physique to develop immune reminiscence. “But it surely offers safety for a time period till it wears off,” he mentioned. The same kind of therapy was used to assist protect immunocompromised patients against COVID.
Domachowske, who additionally led the hospital’s COVID-19 vaccine trial for kids, expects a greenlight from regulators in time to have nirsevimab out there by the following RSV season within the fall. It has already been approved in Europe.
Lengthy journey to an efficient therapy
When Meany’s daughters bought their injections in January 2015, they had been the primary infants on this planet to obtain it, in accordance with AstraZeneca.
Domachowske, a Meany household pal, mentioned giving the dual infants safety towards RSV was a big second after researchers had struggled for years to discover a therapy to stop RSV. Again within the Sixties, a different treatment, a vaccine candidate, was beneath examine. But it surely made youngsters sicker from RSV – and two infants died from it.
“It actually charged up the incorrect half of the immune system,” Domachowske mentioned.
Progress did not come till 20 years later. In 1998, the FDA OK’d a monoclonal antibody for untimely and high-risk infants. However Domachowske mentioned altering medical tips since then have severely restricted eligibility for this therapy, and, he mentioned, its efficacy wasn’t nice.
“It must be given month-to-month,” Domachowske mentioned. “And it is efficient at stopping hospitalization, not efficient at stopping an infection.”
That is the place the analysis had been caught for years till 2014, when Domachowske attended a medical convention in Argentina. A featured speaker dropped an enormous discovery that a whole lot of RSV analysis centered on the incorrect protein.
“Everyone seems to be sitting there staring with their mouths gaping open like, ‘That is why all of our work hasn’t led to something for many years,” Domachowske mentioned. “It was that spectacular. And you may see the pharma those who had been attending, taking notes, calling their colleagues saying, ‘Cease, cease the work.'”
Not too lengthy later, he injected Meany’s daughters with an improved, longer-lasting monoclonal antibody that protects infants by way of an RSV season with one shot.

The Meany twins had been the primary on this planet to get pictures of nirsevimab throughout early trials once they had been infants. They’d no negative effects and no signs of RSV.
Cheryl Meany
disguise caption
toggle caption
Cheryl Meany

The Meany twins had been the primary on this planet to get pictures of nirsevimab throughout early trials once they had been infants. They’d no negative effects and no signs of RSV.
Cheryl Meany
The dual ladies, Cassidy and Stella, at the moment are 8 years outdated and prefer to compete in ninja warrior contests — they race by way of impediment programs that function ladders, monkey bars and overturned Bosu balls.
Meany mentioned the ladies by no means had issues from the shot and by no means displayed signs of RSV. She is pleased with the position they performed in medical historical past.
“This issues, and this issues for teenagers all over the place, not simply youngsters right here,'” Meany mentioned.
Domachowske mentioned the ladies might have gotten RSV in later seasons after the consequences of the therapy had worn off. However since older kids’s immune methods are stronger, signs weren’t noticeable.
A welcome RSV prevention instrument
Physicians and infectious illness specialists welcome the potential approval of the therapy.
Schaffner of the Nationwide Basis for Infectious Illnesses mentioned if it had been already authorized within the U.S., nirsevimab would’ve helped curb the excessive charge of infections seen this season, one of many worst recent seasons for the disease.
“This latest surge would have been remarkably blunted,” he mentioned.
Dr. Vandana Madhavan, medical director of pediatric infectious illness at Mass Normal for Youngsters mentioned the monoclonal antibody is a big achievement within the struggle towards RSV.
“This can be a big step ahead,” she mentioned.
[ad_2]
Source link