[ad_1]
CNN
—
The producer of eye drops which were linked to an outbreak of great bacterial infections within the US, together with no less than three deaths, didn’t comply with correct protocol to stop contamination of its merchandise, based on an inspection report printed Friday by the US Meals and Drug Administration.
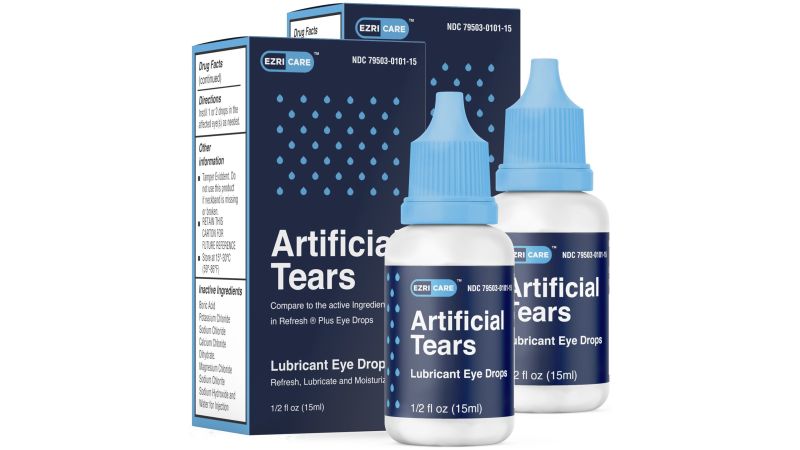
The FDA visited a International Pharma Healthcare facility in India for an inspection that began in mid-February, 2½ weeks after the corporate recalled EzriCare Synthetic Tears as a result of attainable contamination.
On the time of the recall, there have been 55 experiences of antagonistic occasions together with eye infections, everlasting lack of imaginative and prescient and no less than one loss of life with a bloodstream an infection. As of late last month, 68 infections had been recognized in 16 states, based on the US Facilities for Illness Management and Prevention. There have been three deaths, eight instances of imaginative and prescient loss and 4 surgical eye removals reported.
An 11-day inspection of the International Pharma facility resulted in 11 observations by the FDA, together with a “manufacturing course of that lacked assurance of product sterility,” particularly for batches of product that have been manufactured between December 2020 and April 2022 and shipped to the US.
The EzriCare Synthetic Tears product, which is manufactured by International Pharma, is a part of an outbreak of infections from micro organism referred to as Pseudomonas aeruginosa.
This uncommon drug-resistant micro organism can unfold amongst individuals who don’t have signs – and to individuals who haven’t used the attention drops, based on the CDC. The sort of unfold is especially widespread in well being care settings.
“The micro organism can unfold when one affected person carrying the micro organism exposes one other affected person, or when sufferers contact widespread gadgets or when healthcare employees transmit the germs which is why an infection management, like hand hygiene, is so vital,” the company advised CNN in an e mail Monday.
A number of instances within the present outbreak have been recognized in individuals who have been carrying the micro organism with out indicators or signs of medical infections, the CDC stated. These instances have been found by means of screenings at inpatient well being care services that had clusters of infections.
The actual pressure of the micro organism related to this outbreak had by no means earlier than been reported within the US, and associated infections have been recognized at acute care hospitals, long-term care services, emergency departments, pressing care clinics and different outpatient services.
Folks affected by the outbreak reported utilizing completely different manufacturers of synthetic tears, however EzriCare Synthetic Tears was mostly reported.
The FDA inspection of the International Pharma facility is a part of an ongoing compliance matter.
“The FDA’s highest precedence is defending public well being – this contains working with producers to rapidly take away unsafe medication from cabinets when they’re recognized,” the company stated in an e mail Monday. “The FDA continues to observe this situation and is working with the Facilities for Illness Management and Prevention (CDC) and the businesses recalling these affected merchandise. We urge customers to cease utilizing these merchandise which can be dangerous to their well being.”
[ad_2]
Source link